Melanie Lenhardt, senior consultant, and Mohammed Dayazada, head of performance excellence competence centre, Camelot, write about how a changing situation requires adaption of supply chains in the pharma industry
With a projected annual growth rate of 9.2% by 2020 the Gulf Countries are one of the fastest growing pharmaceutical markets in the world. At the same time, the region is battling various challenges such as limited workforce resources, changing regulations as well as limited research and development capabilities.
Local governments, pharma manufacturers and logistics service providers have no alternative but to make decisive changes to their supply chains if they want to capitalise on the significant market opportunities.
This is the result of CAMELOT Management Consultants current market report Blooming and Booming: The Pharmaceutical Market in the Gulf Region.
The majority of the Gulf Countries, comprising mainly the six GCC states Bahrain, Kuwait, Qatar, UAE, Saudi Arabia and Oman plus Iran, are importers of patented pharmaceutical products, with overall imports contributing around 70 to 85% of total consumption. However, certain markets in the Middle East (e.g. Jordan, Egypt, and Saudi Arabia) have a generic drugs manufacturing market, which is continually growing along with the changing demographics in the region.
Prevalence of lifestyle diseases such as diabetes and obesity, population growth and rapid urbanisation are just a few reasons why governments of the Gulf Countries are heavily promoting initiatives in the public and private sector to establish an increasingly self-sufficient pharmaceutical manufacturing base with the following objectives: Reducing dependency on imports, lowering governmental expenditure on healthcare, providing higher caliber jobs for local citizens, increasing availability and quality of healthcare products and services to the population and the diversification of economy and attraction of foreign direct investments
Yet, the local pharmaceutical industry is at a crossroads: limited workforce resources, changing regulations, and limited research and innovative products capabilities are just a few issues the industry is battling in the Gulf Countries. Consequently, governments, manufacturers and Logistics Service Providers (LSPs) have no alternative but to make decisive changes to their supply chains if they want to successfully overcome these challenges and capitalise on the significant manufacturing and distribution opportunities of this rapidly growing market.
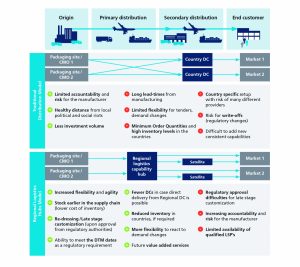
Responding Supply Chain Configurations in GCC States
The majority of the GCC pharmaceutical markets are growing at faster rates than in Western Europe and North America, providing opportunities for international companies. Nevertheless, regulatory, economic, and demographic developments create obstacles within the pharmaceutical supply chain which need to be considered before investments into the region are made. The governments (with regulations and investment in infrastructure), manufacturers and LSPs are the main stakeholders in the pharmaceutical supply chain with different challenges.
Turning the region into an attractive manufacturing base
GCC governments need to attract more foreign investment into the pharmaceutical industry in order to reduce public healthcare costs. The strategy of most GCC states is to reduce oil-dependency by an increasing diversification of the economy. One governmental approach is turning the region into an attractive manufacturing base for national and international companies, which will bring in technological know-how, expertise and resources. GCC governments have established various sponsored incentives schemes as establishment of multiple free zones, beneficial positioning in local and regional governmental tenders and attractive land lease and utility rates, to attract investors for local manufacturing.
For example, Saudi Arabia has a very strong pharmaceutical manufacturing base with 12 major, well-established companies in pharmaceutical clusters in Riyadh, Al Qassim and Jeddah supplying less than a third of the local market. In addition, four major global players including Pfizer and Sanofi have recently completed the construction of production plants within King Abdullah Economic City (KAEC) making it one of the most attractive pharmaceutical clusters for new investors in the region.
However, to capitalise on the economic advantages, the planned pharmaceutical initiatives require a local qualified workforce across different levels, which is currently lacking in the GCC. Several initiatives to reduce this cap and to increase the skill set availability in the pharmaceutical supply chain are taking place, yet at a slow rate.
Logistics infrastructure in the region is moving towards the patient-centric perspective
With the empowerment of the pharmaceutical up and downstream manufacturing base in the GCC, several primary and secondary distribution models are present in the region with a trend towards establishing local distribution centers (instead of the traditional distribution model), in which the products are transported from the manufacturer site to a regional logistics hub (e.g. in UAE) before being allocated to the local distributor (e.g. in Oman).
Countries as the UAE and Oman support the establishment of regional logistics hubs. The UAE is seen as a mature market for distribution due to the established governmental initiatives such as free trade zones focusing on logistics, healthcare and manufacturing. In addition, these initiatives are complimented by storage, distribution and manufacturing regulations focusing on quality, safety and security, all of which are important to pharmaceuticals companies.
However, the main challenge is that GCC countries currently cannot be used as distribution hubs for the Saudi marketthe largest pharmaceutical market in the GCCas Saudi health regulations forbid the import of drugs from countries other than the Country of Origin.
Therefore, Saudi Arabia is currently not suitable to become a regional distribution hub for pharmaceutical products due to the unavailability of free trade zones which are a crucial requirement as they ensure that no duties are applied on the products until they are received by the final destination. Nevertheless, over the last four years, the Saudi government has initiated several projects to establish Saudi Arabia as a regional logistics hub for varied industries and improve on the logistics efficiency in the Kingdom. One of the key initiatives is to study the establishment of economic zones in multiple locations with trade, financial and corporate benefits and incentives.
Another initiative is to revamp the country´s port infrastructure and optimise import and export processes in line with global and regional benchmarks. Moreover, government-owned industrial city authorities as Royal Commission and MODON are preparing for the upcoming rail connectivity and the establishment of multi-modal logistics hubs across the Kingdom, which will be an enabler for attracting, diversified industries and specialized LSPs. When these initiatives have materialized, Saudi Arabia will capture a significant market share of the pharmaceutical distribution in the GCC and further raise the growth of the pharmaceutical market in the Middle East.
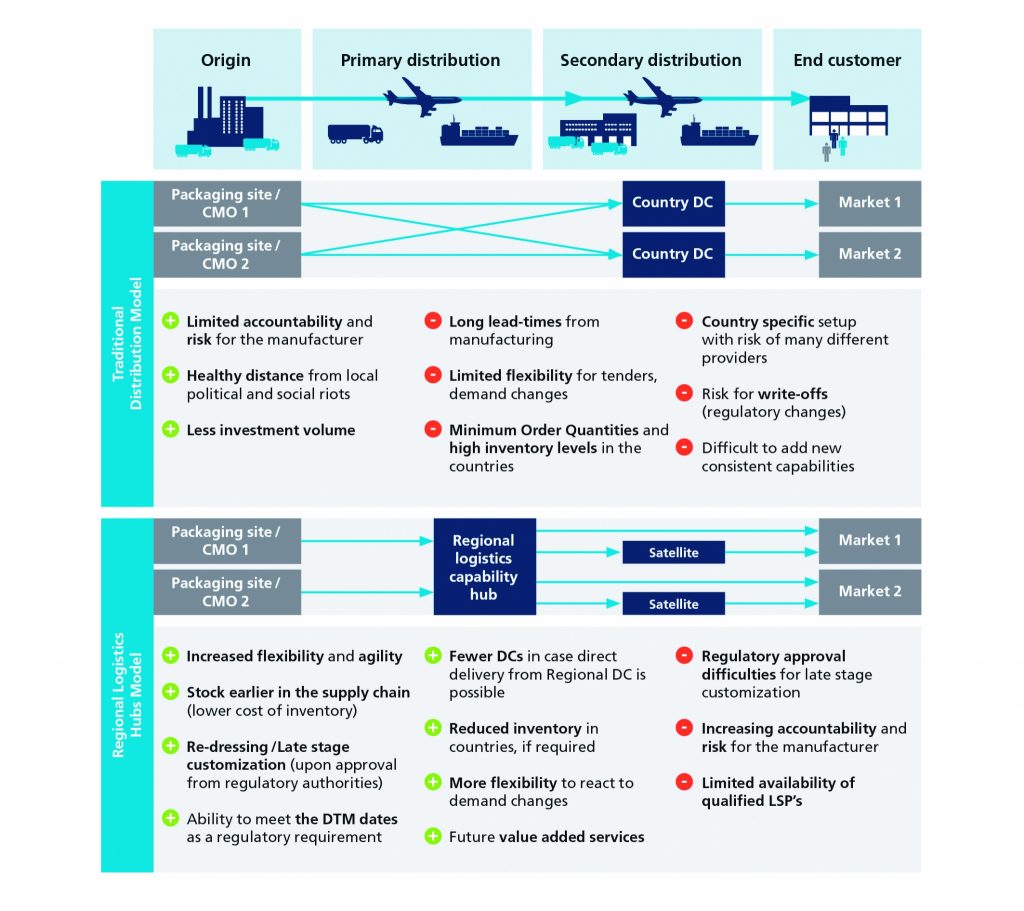
Getting ready for additional value added services by LSPs
Increasing cost pressure, regulatory complexity and demand fluctuations require comprehensive modification throughout the pharmaceutical supply chain, which have for years been influenced mainly by the demand for high delivery reliability and high delivery readiness. Therefore, in the Gulf region, value-added services such as secondary packaging and postponement become increasingly important, says Mohammed Dayazada, co-author of the study and Competence Center Lead at CAMELOT in Dubai.
Both describe logistics and manufacturing concepts in which a certain level of semi-finished inventory is stored temporarily until a customer order is received, at which point the product is finalized. Within the secondary packaging strategy it is possible to adjust packaging size, leaflets and adding stickers or 2D coding to the product. In comparison, the postponement strategy offers even more customizing and flexibility allowing changes to the product, packaging and labeling at a later stage of the supply chain.
However, beside the benefits from value added services, pharmaceutical manufacturers and LSPs are facing certain obstacles hindering its implementation. For example, in the GCC postponement strategy is regulated by requiring a Good Manufacturing Practice (GMP) license, which can be difficult to obtain, whereas the regulations behind secondary packaging are changing constantly leading to uncertainties.
This means, that only a limited number of LSPs offer these VAS in the GCC. In addition, as VAS are a rather new offering in the region, the qualification of the workforce and the quality of service has not yet reached a satisfying level. Therefore, pharmaceutical companies are working closely with LSPs and Health Authorities to close these gaps through knowledge exchange, partnerships and collaborations.
Iranian market will be a game changer in the Middle East region
Another key result of the study is that the opening of the Iranian market, which is the worlds 17th biggest economy with a population of 80 million, will be a significant game changer for the region.
Iran is one of the last untapped pharmaceutical markets in the region. The Iranian market provides pharmaceutical manufacturers and logistics service providers numerous economic benefits and profitable opportunities, says Melanie Lenhardt, co-author of the study and Senior Consultant at CAMELOT in Dubai.
With regards to the above mentioned three key components of the pharmaceutical supply chain (manufacturing, distribution and value added services) the question occurs whether Iran offers the right prerequisites to become a leading pharmaceutical market in Middle East. Restrictions on bank payment methods in particular resulted in a shortage of imported drugs and medical equipment. This did lead to the fact, that the existing pharmaceutical companies are out-of-date with less than 5% of the manufacturing plants complying with international standards.
Therefore, the Iranian government announced a new project with the aim to attract foreign direct investments and to highlight the attractiveness of the market: the development of an Industrial Pharmaceutical City on 176 hectares of land only 65km away from Tehran providing benefits including tax exemptions and export facilitation.
In comparison, Saudi Arabia, the largest pharmaceutical market among the GCC states, remains to have restrictive regulations which limit the opportunities for regional distribution and manufacturing. The near future will show whether Saudi Arabia as a rather slowly opening market will offer more attractive opportunities and benefits for investors than the fast opening Iranian market.
About the authors
Mohammed Dayazada is Head of Performance Excellence Competence Center at CAMELOT Management Consultants MEA and is a regional pharma expert located in Dubai. He gained profound experience within global pharmaceutical companies in the area of supply chain optimization and network design. He had several engagements with governmental institutions to develop pharmaceutical clusters and attract pharmaceutical investors to the region.
Melanie Lenhardt is Senior Consultant at CAMELOT Management Consultants MEA located in Dubai. She gained international consulting and pharma industry experience with focus on supply chain management and planning.
The Blooming and Booming: The Pharmaceutical Market in the Gulf Region study was conducted in cooperation with the Association Supply Chain Management, Purchasing and Logistics (BME), and is available at www.camelot-mc.ae.

